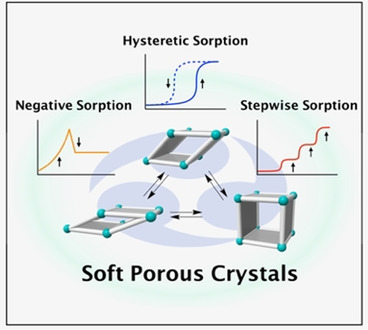
Illsutration of adsorption phenomena in soft porous crystals see original work here
More or less by accident I discovered that under certain conditions porous solids can contract their pores during the adsorption process and release adsorbed gas from their pore as a consequence of reduced pore diameter. The phenomenon was concequently called „negative gas adsorption“. S. Krause, V. Bon, I. Senkovska, U. Stoeck, D. Wallacher, D. M. Többens, S. Zander, R. S. Pillai, G. Maurin, F. o.-X. Coudert, S. Kaskel, A pressure-amplifying framework material with negative gas adsorption transitions. Nature 2016,532 (7599), 348-352. To date most adsorption phenomena in soft porous materials are discovered by accident and fundamental understanding of the properties and structural motives that govern these non-classical properties are still lacking. With the help of computational methods we try to explore criteria that efine such „soft adsorption processes“.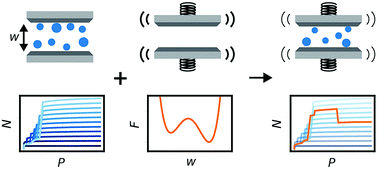
Toy-model to represent pore expansion and contraction in soft porous materials. see original work
J. D. Evans, S. Krause, S. Kaskel, M. B. Sweatman, L. Sarkisov, Exploring the thermodynamic criteria for responsive adsorption processes. Chemical Science 2019,10 (19), 5011-5017. A key feature in these systems is the complex energy landscape that depends on many different variables of the solid but also liquid phase. Brining both together is a key step in designing novel adsorption properties and utilize them for applications and technologies that currently do not exist.